Flip over a medicine strip, and you will see familiar two or three uppercase letters quietly sitting next to a medicine’s name. SR. ER. XR. Most people flip the strip, swallow the pill, and move on. But those abbreviations? They are not just simple letters.
They are engineering decisions.
They decide how fast a drug hits your system, how long it stays effective, and sometimes, whether it needs to be taken once a day or thrice. In other words, those letters can shape your treatment outcome without you even noticing.
For this story, I spoke to two doctors, a manufacturer we source medicines from, and the lab analyst who tests every batch we sell. Because understanding these cryptic codes is key to understanding what your medicine is actually doing inside your body. Read this blog and you will never ignore your medicine strip the same way again.
Table of Contents
What Are Modified-Release Tablets?
These are a special type of medicine designed to delay the release of the active ingredient after entering the body. Unlike regular tablets, they do not act all at once. Instead, they are engineered to deliver a steady effect over time, reducing side effects and cutting down how often you need to take them. It’s a smarter way to dose, with the same medicine working longer and more gently.
Also Read: What is the best time to take my medicine?
Types of Modified Release (MR) Tablets
1- Extended Release (ER or XR)
Think of it like a childhood candy something like Alpenliebe or Kacha Bite. You did not chew it all at once; you let the flavor melt slowly. That is exactly what ER medicines are designed to do. They release their active ingredient gradually, stretching the effect over 24 hours. The result? One pill, once a day. No constant re-dosing.
2- Sustained Release (SR)
If you have ever watched a mukbang video, you might have observed the pattern: the eater starts strong, then slows down. SR tablets work similarly. The medicine kicks in quickly with a higher dose upfront, but the release slows down over time. This creates a longer-lasting effect typically 8 to 12 hours without overwhelming the body all at once.
3- Controlled Release (CR)
Imagine a watchman stationed on your terrace, assigned to open the water tank valve every hour and release exactly one liter. That is CR medicine in a nutshell. It delivers a fixed amount of the drug at controlled intervals, ensuring a steady effect throughout the day nothing more, nothing less.
4- Prolonged Release
Think of watering a plant, not with a sudden splash, but with an ice cube that melts slowly, giving just enough water over time. That’s what prolonged-release medicines do. Instead of flooding your system with a full dose at once, they release the drug gradually, maintaining a steady level in your body. Often, one dose a day is enough.
Globally, these terms have distinct meanings. In India, though, they are often used interchangeably. All of them point to one idea: medicines that last longer in the body. Which raises a question, are they interchangeable? That takes us to the next section.
Also Read: What is Really Inside Your Medicine?
Can you swap one modified-release drug for another?
Doctors I spoke to said no very firmly. But when pressed, their answers circled around disease names and a familiar refrain: “Because the doctor wants a specific effect and drug concentration in the patient’s body.” No one was able to clearly articulate the difference between these formulations. They simply repeated: “We use them in chronic patients.”
It left me wondering, do doctors really understand modified-release tablets?
So I asked someone else. Our lab analyst and a manufacturer we source from. Both said the same thing: these medicines are largely interchangeable. Because at their core, modified-release tablets serve one purpose: to stretch out the medicine’s effect over time.
Take Metoprolol succinate, for example. The test report shows how it dissolves, slow and steady. In the first hour, it releases just 12–20% of its active ingredient. By the 20th hour, it has quietly delivered nearly 94%. The medicine is released at a controlled, consistent manner. It is not about how fast it works, but how long it lasts.

Is there a difference between modified-release tablets made for India and those made for the US?
Turns out, yes. While speaking to doctors, our lab analyst, and a manufacturer, one thing stood out: drugs made in India for the US market are not the same as those made for Indian patients. Surprised? So were we.
Dhruv and I dug into the pharmacopoeias, the official drug standards. The US Pharmacopeia (Image 1) lays out specific dissolution parameters for many modified-release tablets. The Indian Pharmacopoeia? Not quite. It simply says dissolution can vary by manufacturer (Image 2). It is a bit like Pepsi. The one bottled in India does not taste quite like the one in the US.
Image 1 (Source: United States Pharmacopeia (USP 48–NF 43), Monograph of M. Published in 2020. Retrieved in 2025 from- USP 48 – NF 43 Monograph of M)
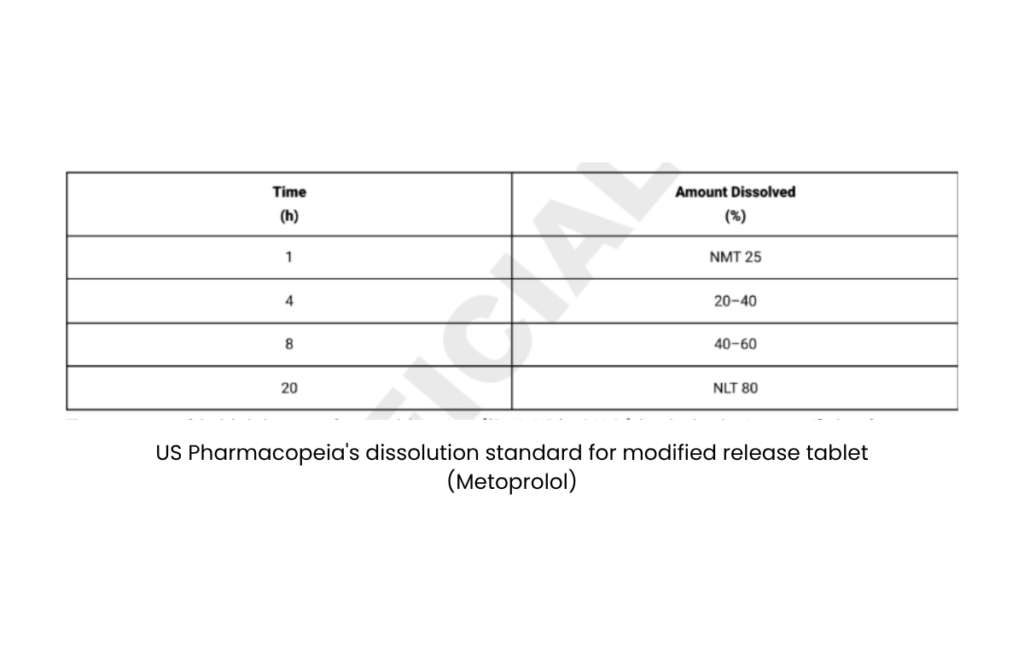
Image 2: (Source: Indian Pharmacopoeia (2022), Volume 2 – Monograph of Metoprolol Succinate Prolonged Release (PR). Published by Indian Pharmacopoeia Commission)

But what if your tablet has no letters?
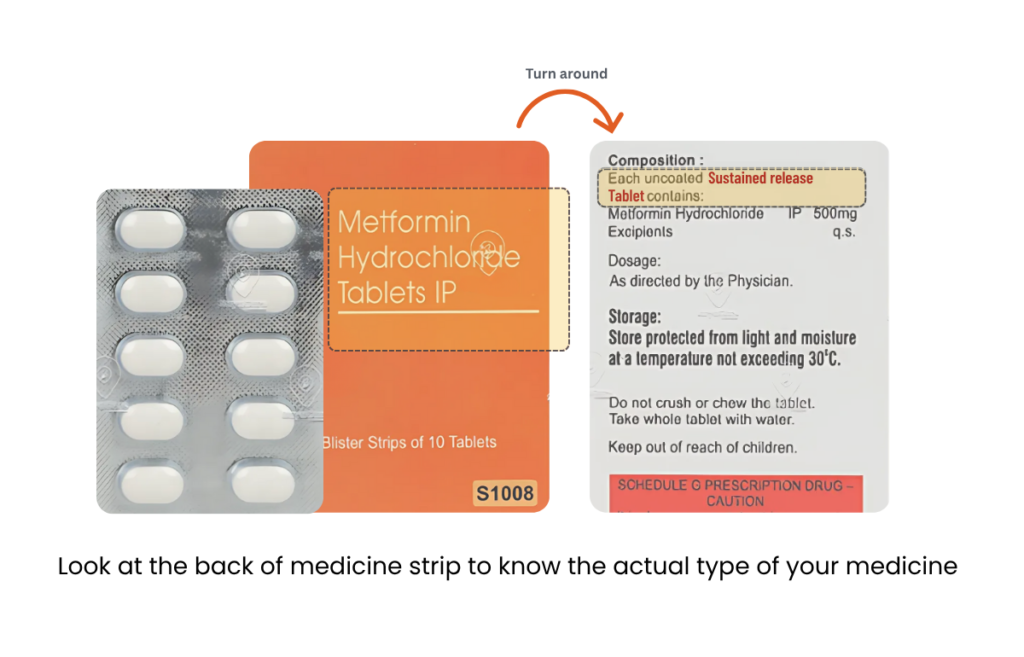
Not every pill comes with labels like ER, SR, PR, or CR. But here is the trick: if there is no label on the front, flip it over. Check the back of the strip. If it says nothing about “extended,” “controlled,” or “sustained” release then It is an immediate-release tablet. That means it kicks in fast and fades out just as quickly.
This one does not show any modified release letters up front. But flip it over, and boom, it says “Sustained Release” on the back. So yeah, it is a modified release medicine. Just hidden in plain sight.
Also Read: Taking Supplements Without a Doctor? Why It Could Be Risky And What to Do Instead
Importance of Modified Release Tablets
1- Low Intake Frequency
Modified release tablets are built for convenience. By delivering the drug slowly over time, they reduce how often you need to take your medicine, sometimes to just once a day.
2- Lower Risk of Side Effects
Because the medicine is released in smaller amounts, it clears from the body more smoothly. That means fewer spikes, less accumulation, and often fewer side effects.
3- Longer Therapeutic Effect
Rather than flooding the system all at once, these tablets release a steady amount of active ingredients. This ensures a consistent therapeutic effect without overwhelming the body.
4- Reduces Dose Dumping
Unlike immediate-release drugs that release everything at once, MR tablets are controlled by design. This reduces the risk of dose dumping a sudden spike in drug levels in blood that can cause side effects or toxicity.
Disadvantages of Modified Release Tablet
1- Difficult to Split
Modified-release tablets are designed to release the drug gradually. Split them, and that control vanishes, delivering the full dose all at once. The entire point is lost.
2- Difficult to Adjust the Dose
Because these medicines stay in the body longer, changing the dose is not as simple as skipping or adding a pill. If your doctor wants to adjust your dosage, they will likely revise your schedule for the following day, not immediately.
Conclusion
More than you think. Those small uppercase letters- ER, SR, CR and PR are not just alphabets on your medicine strip. They are silent cues about how your drug behaves, and how your body responds. Modified release means one dose a day, fewer side effects, and a smoother ride. Immediate release means spikes, crashes, and the need for multiple reminders. And yet, for all their engineering precision, modified-release tablets are often misunderstood, even by those who prescribe them. Doctors insist they are not interchangeable. Manufacturers quietly admit they mostly are. In a country where taking pills is common but pharmaceutical literacy is low, this matters. Because the tiny letters on your tablet may be shaping your treatment more than the name printed next to them.
Mahak Phartyal completed her bachelor’s in pharmacy from Veer Madho Singh Bhandari Uttarakhand Technical University. She previously worked as a Medical Writer at Meril Life Sciences, where she wrote numerous scientific abstracts for conferences such as India Live 2024 and the European Society of Cardiology (ESC). During her college years, she developed a keen research interest and published an article titled “Preliminary Phytochemical Screening, Physicochemical and Fluorescence Analysis of Nyctanthes arbor-tristis and Syzygium cumini Leaves.”












Your sentences move with quiet grace, producing reflective momentum. Ideas unfold naturally, fostering thoughtful engagement, layered understanding, and a meditative connection between reader and prose.
This article is really insightful! I never knew those tiny letters on my medicine strip held so much meaning about how the drug works in my body.
I respect your work, thankyou for all the good articles.